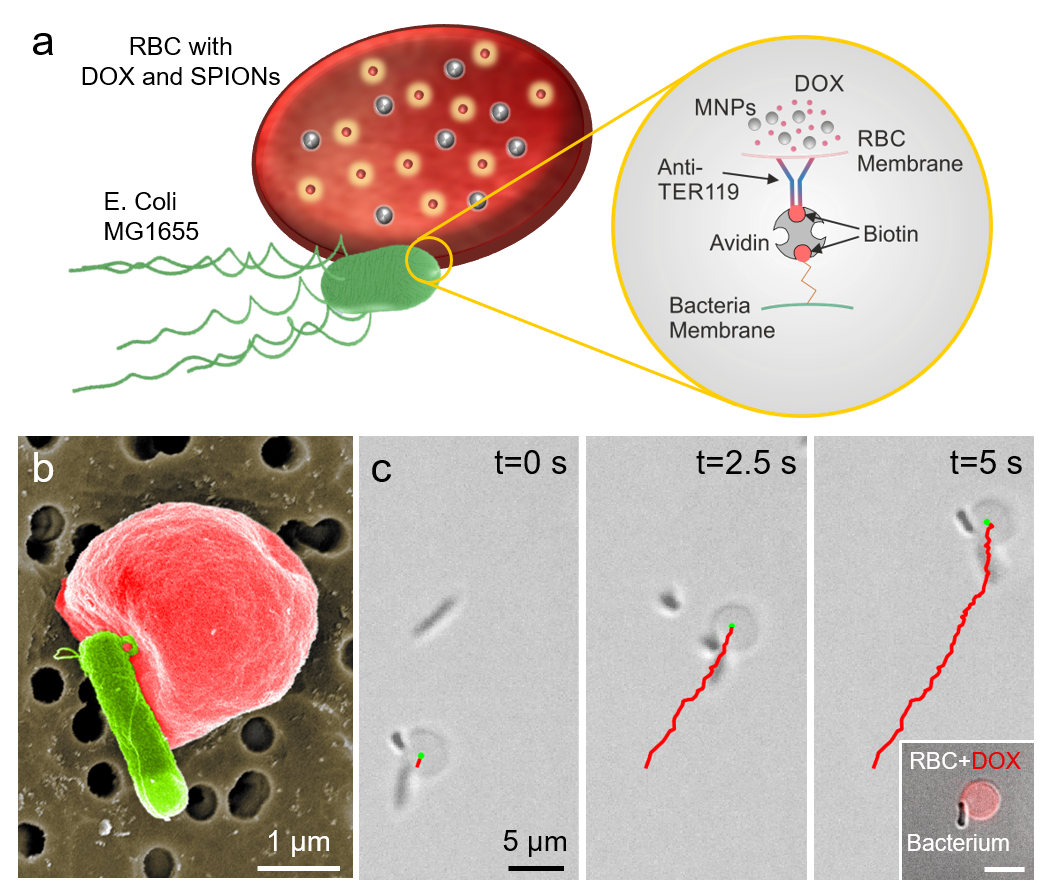
A. RBC microswimmers are composed of an RBC, loaded with drug molecules and superparamagnetic iron oxide nanoparticles, bounds to a motile bacterium via biotin-avidin-biotin binding complex. B. Scanning electron microscope image (pseudo-colored red: RBC; pseudo-colored green: bacterium) of an example RBC microswimmer with an attached bacterium. C. Example 2D propulsion trajectories of RBC microswimmers via bacteria over time. The inset displays a bacterium attached to an RBC, loaded with DOX molecules. Scale bar represents 5 $\mu$m.
Bacteria-powered biohybrid microswimmers have recently shown to be able to actively transport and deliver cargos encapsulated into their synthetic constructs to specific regions locally. However, use of synthetic materials as cargo carriers can result in inferior performance in load-carrying efficiency, biocompatibility, and biodegradability, impeding clinical translation of biohybrid microswimmers. In this project, we report construction and external guidance of bacteria-driven microswimmers using red blood cells (RBCs, erythrocytes) as autologous cargo carriers for active and guided drug delivery. Multifunctional biohybrid microswimmers were fabricated by attachment of RBCs (loaded with anti-cancer doxorubicin drug molecules and superparamagnetic iron oxide nanoparticles (SPIONs)) to bioengineered motile bacteria, E. coli MG1655, via biotin-avidin-biotin binding complex. Autonomous and on-board propulsion of biohybrid microswimmers were provided by bacteria, and their external magnetic guidance was enabled by SPIONs loaded into the RBCs. Furthermore, bacteria-driven RBC microswimmers displayed preserved deformability and attachment stability even after squeezing in microchannels smaller than their sizes, as in the case of bare RBCs. In addition, an on-demand light activated hyperthermia termination switch was engineered for RBC microswimmers to control bacteria population after operations. RBCs, as biological and autologous cargo carriers in the biohybrid microswimmers, offer significant advantages in stability, deformability, biocompatibility, and biodegradability over synthetic cargo-carrier materials. The biohybrid microswimmer design presented in this study transforms RBCs from passive cargo carriers into active and guidable cargo carriers toward targeted drug and other cargo delivery applications in medicine.
A curious hybrid of a red blood cell and an E. coli bacterium could make an ideal transporter for carrying drugs inside the body https://t.co/w5IJo7mVq7 pic.twitter.com/o0jhtBvc1C
— New Scientist (@newscientist) April 26, 2018
